|
8/31/2023 0 Comments Endo pharma things to look for
The trusted Legacy Cannabis + Social Equity company We envision a world where human health is transformed through the accessible and widespread. As a specialty pharmaceutical company, were motivated by a strong sense of purpose to find better ways to meet unique medical needs. With a sterile manufacturing facility located in Rochester, Michigan, Par Sterile Products possesses an unparalleled reputation for high-quality products and an impeccable record of regulatory compliance. Endo Industries 177 followers on LinkedIn. Par Sterile Products develops, manufactures and markets a broad portfolio of branded and generic aseptic injectable products. Our focus is on therapeutically equivalent ANDA-based products, and others on a case-by-case basis. Par Pharmaceutical specializes in modified-released oral solid dosage forms as well as non-oral dosage forms, such as nasal sprays, inhalers, patches and other alternative drug delivery platforms. Endo International plc is an American Irish-domiciled generics and specialty branded pharmaceutical company that generated over 93 of its 2017 sales from. Endo Pharmaceuticals Activity Issues the FTC should be focused on: consolidation in the generic customer base and the effects on quality and certainty of supply for 90 of US Rx Liked by Anand. We offer high-quality, cost-effective products with the highest level of customer service. Last week, shares of pharma giant Johnson & Johnson JNJ surged after it announced the launch of its large-scale, multi-country phase III study (ENSEMBLE) for its COVID-19 vaccine candidate, JNJ-78436735.Par Pharmaceutical, a wholly owned Endo business, is a highly focused generic pharmaceutical company dedicated to delivering quality medicines to patients in need through excellence in innovation, development, manufacturing, and commercialization. The pharma and biotech companies are running a race against time to develop a vaccine and combat the contagion. The companys Endo Pharmaceuticals segment. As part of the bankruptcy, Endo’s creditors have agreed to take over the business’s assets in exchange for extinguishing over 5. The race for the development of a vaccine for COVID-19 has intensified in the last couple of months, as a second wave of the pandemic is likely to grip the western countries. provides specialty healthcare solutions in the United States and internationally. Endo Pharmaceuticals, an Endo International company, develops and commercializes nonsurgical therapies in men’s health, orthopedics, and other specialties. Following a special, first of its kind in the opioid sector request from the US Food and Drug Administration (FDA), Endo Pharmaceuticals (NASDAQ:ENDP) has agreed to take Opana ER off shelves. What happened Shares of Endo Pharmaceuticals ( ENDP 2.36) were on the rise. 
Shares of Endo have lost 32.8% in the year so far compared with the industry’s 8.5% decline. Endp stock View todays Endo International PLC stock price and latest ENDPQ. 
Data from this study are likely to support the regulatory submissions for licensure of the vaccine in the United Kingdom, the EU and other countries. Shares also gained last week after the company announced that it has initiated the first phase III study in the United Kingdom evaluating the safety, efficacy and immunogenicity of NVX-CoV2373. Novavax’s shares gained 10.8% on the news. We offer an extensive range of prescription products across a wide range of theraputic categories. headquarters in Malvern has launched a marketing campaign to promote Xiaflex. Endo International based in Dublin, Ireland, with a U.S. Interim data from these studies are expected before the end of 2020. Health Canada approves Endo’s anti-seizure pills - Pharmaceutical Technology News JHealth Canada approves Endo’s anti-seizure pills Xcopri works by reducing repetitive neuronal firing by inhibiting voltage-gated sodium currents, leading to a reduced occurrence of seizures. Endo International has adopted a marketing campaign for its drug to treat Peyronies disease, a painful and rare condition in men. The specialty pharmaceutical company, Endo International plc, has agreed to acquire the privately held specialty pharmaceutical company, Par Pharmaceutical Holdings, Inc., for 8.05 billion, including assumption of Par debt. 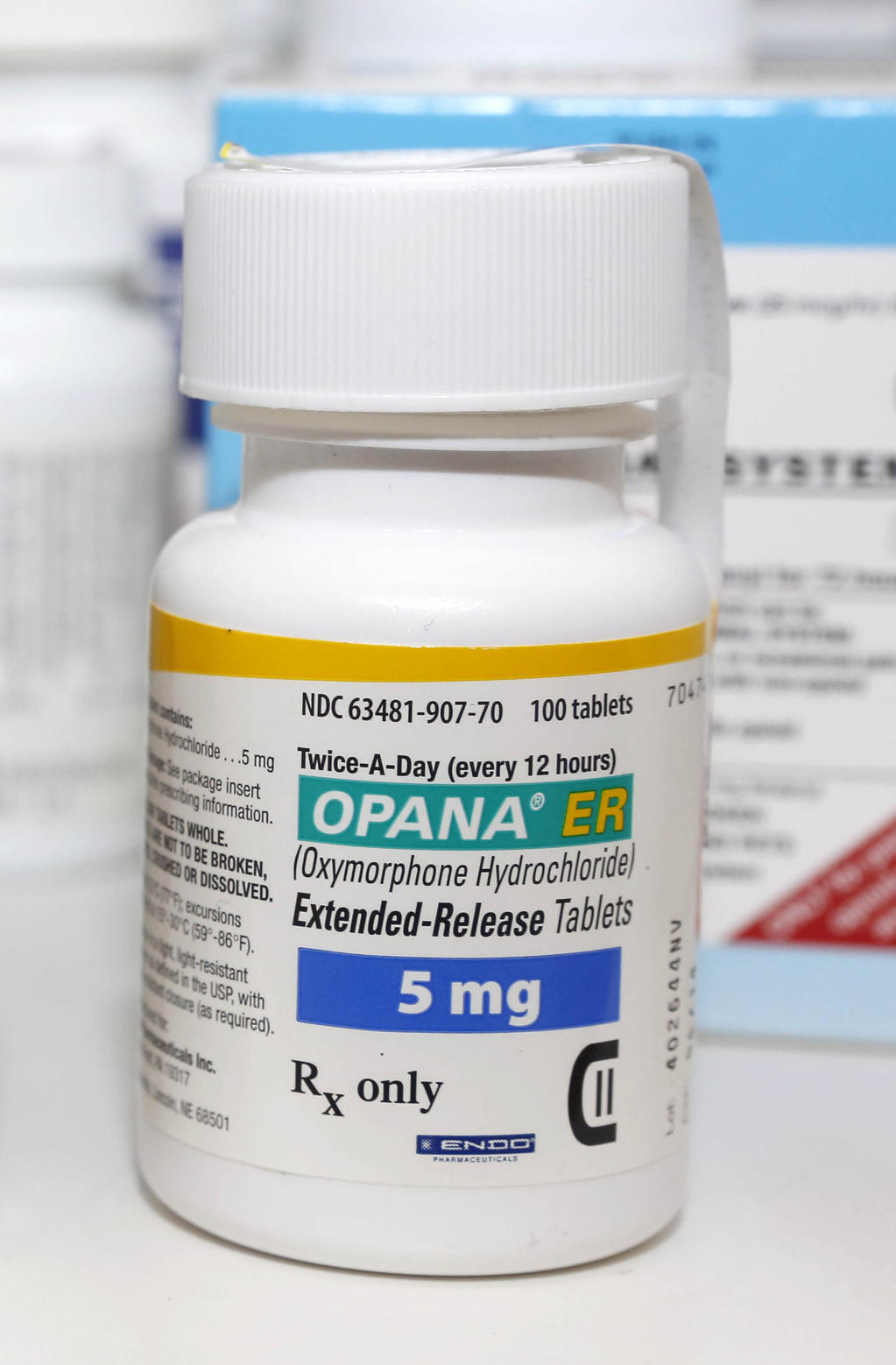
A phase IIb study to assess efficacy began in South Africa in August. DCAT Value Chain Insights (VCI) takes an inside look. A phase II study to evaluate the safety and immunogenicity of NVX-CoV2373 began in August 2020 in the United States and Australia building on positive phase I results and expanding to include older adults. We note that NVX-CoV2373 is currently in multiple phase II studies.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
